40 Hz Light Flickering Alleviates Chronic Pain via Adenosine Signaling
SNIPPET: 40 Hz flickering light alleviates chronic inflammatory and neuropathic pain in mice by activating a direct retina-to-central amygdala (CeA) neural pathway that increases extracellular adenosine levels. This adenosine acts through A2A receptors to suppress nociceptive responses and may erase pain memory traces — offering a non-invasive, drug-free alternative to opioids and NSAIDs for chronic pain management.
THE PROTOHUMAN PERSPECTIVE#
Chronic pain affects over 1.5 billion people globally. The current pharmacological toolkit — NSAIDs that erode your gut lining, opioids that hijack your reward circuitry — is failing. Not slightly underperforming. Failing. We've spent decades looking for better molecules to swallow when the signal might have been sitting in the visible spectrum all along.
This research from Cell Research maps a previously unknown optical pathway directly from the retina to the central amygdala, the brain's pain-processing and aversive memory hub. The implications for human performance optimization are significant: if validated in humans, 40 Hz flicker could become the first light-based, zero-side-effect intervention for chronic pain that actually has a mechanistic explanation. Not another vague "photobiomodulation helps inflammation" hand-wave. A specific circuit. A specific neurotransmitter. A specific receptor subtype.
For those of us tracking non-invasive neuromodulation, this is the kind of preclinical data that earns attention — not because it promises everything, but because it explains something.
THE SCIENCE#
What Is 40 Hz Light Flickering and Why Does It Matter?#
Forty-hertz light flickering is a visual stimulus pulsing at gamma frequency — 40 cycles per second — delivered at controlled luminance. It matters because gamma-frequency entrainment has emerged as one of the most promising non-invasive neuromodulation strategies of the past decade, initially studied for Alzheimer's disease pathology clearance and now extending into sleep, pain, and glymphatic function. The data here is specific: in mouse models, 40 Hz flickering at 2000 lux significantly reduced mechanical hyperalgesia in both inflammatory (CFA) and neuropathic (SNI) pain models, with effects persisting more than 6 hours after stimulation ceased [1]. The US FDA designated the 40 Hz flicker paradigm (GENUS equipment) as a "breakthrough device" in 2021, and the research group behind this pain study has now published three separate papers establishing adenosine as the mechanistic link [3].
The Retina-CeA Pathway: A Direct Line from Eyes to Pain#
Here's where this gets interesting — and where I start paying closer attention.
The researchers didn't just show that shining a flickering light at mice reduces pain behavior. They dissected the circuit. Using anterograde transsynaptic tracing (AAV1-hSyn-Cre injected intravitreally), they identified retinal ganglion cells (RGCs) that project directly to the central amygdala. Not through the visual cortex. Not through the thalamus. A direct retina-to-CeA connection [1].
This is the critical finding. Circuit-specific manipulations confirmed that optogenetic activation of these retinorecipient CeA neurons was sufficient to produce analgesia, and silencing the pathway with tetanus toxin (TeNT) abolished the analgesic effects of 40 Hz flickering [1]. Both sufficiency and necessity demonstrated in the same study. That's a high bar for a circuit claim, and they cleared it.
The n was small — 6 mice per group in the optogenetic experiments — and I'd want to see this replicated independently before treating it as settled neuroscience. But the experimental design is clean.
Adenosine: The Molecular Mechanism#
The mechanistic story connects to the group's earlier work on 40 Hz flickering and sleep. In 2024, they published in Cell Research demonstrating that 40 Hz flicker increases extracellular adenosine in the primary visual cortex (V1) via ENT2 (equilibrative nucleoside transporter-2), generated through AMPK-associated energy metabolism pathways in cortical neurons [3]. The adenosine increase was frequency-dependent — maximal at 40 Hz, absent at 20 Hz and 80 Hz — and intensity-dependent, peaking at 4000 lux.
The new pain study extends this to the CeA specifically. 40 Hz flickering significantly increased extracellular adenosine levels in the CeA, and pharmacological blockade of ENT prevented this increase and abolished analgesia [1]. When the researchers directly infused adenosine into the CeA, it phenocopied the light-induced pain relief. Both interventions required A2A receptor signaling to work.
Let me be precise about what this means. The pathway is: 40 Hz light → retinal ganglion cells → CeA neurons → neuronal adenosine generation via AMPK/energy metabolism → ENT2-mediated adenosine efflux → extracellular adenosine → A2A receptor activation → suppressed nociceptive responses.
That's a lot of steps, and each one was experimentally validated. I'm genuinely impressed by the thoroughness.
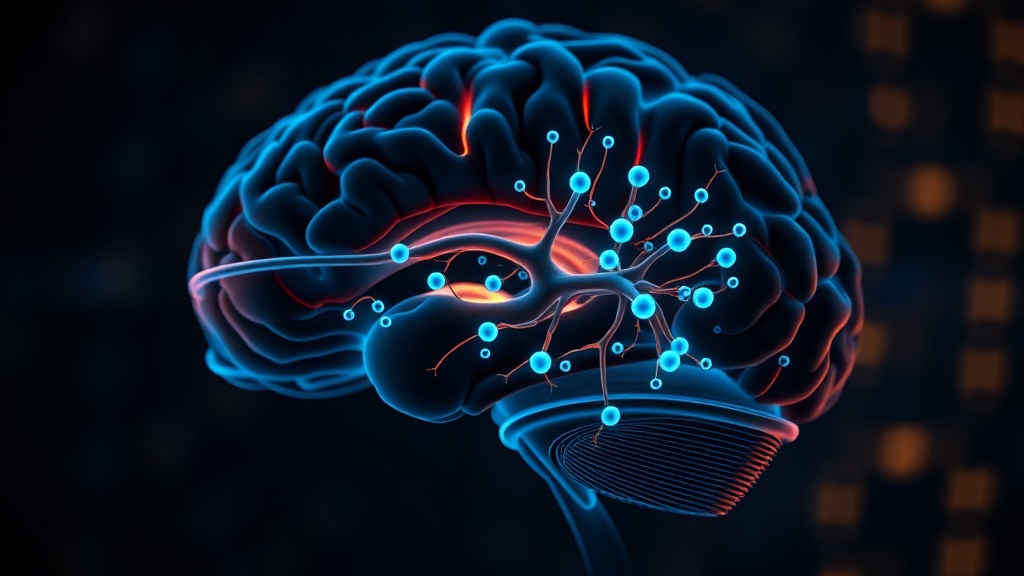
Pain Memory Erasure: The Most Provocative Claim#
Now here's where I want to push back slightly. The study also claims that 40 Hz flickering can "erase pain memory" by destabilizing hyperalgesia traces in the CeA, mirroring reconsolidation processes [1]. The concept is sound — memory reconsolidation requires protein synthesis, and disruption during the reconsolidation window can weaken or eliminate the memory trace. This has been demonstrated for fear memories. Extending it to pain memory in the CeA is a logical step.
But "pain memory erasure" is a strong claim. The behavioral readout (paw withdrawal threshold) doesn't distinguish between active analgesia and genuine memory destabilization. I'd want to see molecular markers of reconsolidation disruption — protein synthesis inhibition controls, zif268 expression, that kind of verification — before accepting this interpretation fully. The data suggests it. It doesn't yet prove it.
Supporting Evidence: The Adenosine Story Keeps Building#
A separate study published in January 2026 in Communications Biology demonstrated that 40 Hz flicker preconditioning protects against ischemic optic neuropathy (NAION) through — again — adenosine signaling, specifically via A1 receptors and ENT-mediated transport [2]. Three days of 40 Hz flicker before ischemic insult reduced retinal ganglion cell loss and preserved visual function for at least 4 weeks. The neuroprotection was frequency-dependent: effective at 40 Hz, absent at 20 Hz and 80 Hz.
This convergence across three studies (sleep, pain, ischemic neuroprotection) on the same adenosine mechanism at the same frequency is compelling. It suggests 40 Hz isn't an arbitrary number — it maps onto a specific neuronal metabolic threshold for adenosine generation.
Analgesic Efficacy by Flicker Frequency (2000 lux, CFA Model)
COMPARISON TABLE#
| Method | Mechanism | Evidence Level | Cost | Accessibility |
|---|---|---|---|---|
| 40 Hz Light Flickering | Retina-CeA adenosine signaling via ENT2/A2A receptors | Preclinical (mouse models, strong mechanistic data) | Low (LED device) | High potential — non-invasive, home-use |
| NSAIDs | COX-1/COX-2 inhibition, prostaglandin reduction | Extensive human RCTs; limited chronic efficacy | Very low | Over-the-counter |
| Opioids | µ-opioid receptor agonism | Extensive human RCTs; high efficacy, high risk | Low–moderate | Prescription only; addiction risk |
| Transcranial Direct Current Stimulation (tDCS) | Cortical excitability modulation | Mixed human RCTs; small effect sizes | Moderate ($200–$500 device) | Home-use devices available |
| Red/NIR Photobiomodulation | Mitochondrial cytochrome c oxidase activation | Moderate human evidence; mechanism partially defined | Moderate ($100–$2000) | Home-use devices widely available |
| Spinal Cord Stimulation | Gate control theory; dorsal column modulation | Strong human evidence for neuropathic pain | Very high ($30K–$50K+) | Surgical implant; specialist only |
THE PROTOCOL#
Important caveat: This protocol is extrapolated from preclinical mouse data. No human clinical trial for 40 Hz flicker and chronic pain has been published. If you choose to trial this based on current evidence, treat it as experimental self-quantification, not established therapy.
Step 1: Source the correct hardware. You need a flicker device capable of delivering precisely 40 Hz (not approximate — 38 Hz or 42 Hz will not produce the same neurochemical response). Consumer "gamma entrainment" devices exist, but verify the actual output frequency with an oscilloscope or photodiode if possible. The study used 2000 lux at the eye. Most commercial gamma flicker devices deliver substantially less. Wavelength matters less than frequency and intensity here — the study used white light.
Step 2: Establish baseline pain metrics. Before starting, quantify your pain using a validated scale: the Visual Analog Scale (VAS, 0–10) or the Brief Pain Inventory. Record for at least 3 days before beginning flicker sessions. Without a baseline, you cannot distinguish placebo response from genuine effect.
Step 3: Begin daily sessions at 1 hour. The mouse study found that 1-hour and 4-hour daily sessions produced comparable analgesic effects [1]. Start with 1 hour per day. There's no evidence that longer sessions improve outcomes, and 1 hour is the minimum effective dose tested.
Step 4: Position the light source correctly. The light must reach the retina — this is not a transcranial or skin-applied intervention. Sit facing the flickering light source at a distance that delivers approximately 2000 lux to your eyes. Keep eyes open (blinking normally is fine). The retina-CeA pathway requires direct retinal stimulation.

Step 5: Maintain daily consistency for at least 5–7 days. In the CFA mouse model, significant analgesia emerged after several days of daily stimulation. Analgesic effects persisted for more than 6 hours after each session [1]. Schedule your session in the morning or early afternoon — the adenosine buildup may promote sleepiness (this group's earlier work showed 40 Hz flicker promotes sleep via cortical adenosine [3]).
Step 6: Track and reassess. Record VAS scores before each session, immediately after, and at 2, 4, and 6 hours post-session. If no improvement after 10 days, discontinue. If improvement occurs, note whether it persists overnight or requires daily reinforcement.
Step 7: Monitor for photosensitivity. Individuals with epilepsy or photosensitive conditions should NOT attempt 40 Hz flicker without medical supervision. The 40 Hz frequency is within the range that can trigger photosensitive seizures in susceptible individuals. This is non-negotiable.
Related Video
What is the retina-CeA pathway and why does it matter for pain?#
The retina-CeA pathway is a direct neural connection from retinal ganglion cells in the eye to the central amygdala, a brain region critical for processing aversive experiences and pain. This study identified it as the conduit through which 40 Hz flickering light produces analgesia in mice [1]. It matters because it provides a specific, targetable circuit — not a diffuse whole-brain effect — that links visual input directly to pain modulation.
How does adenosine reduce pain in this context?#
Forty-hertz flickering light increases extracellular adenosine levels in the CeA through ENT2-mediated transport. This adenosine activates A2A receptors on CeA neurons, suppressing nociceptive (pain-signaling) responses [1]. Adenosine is the body's endogenous neuromodulator — the same molecule that accumulates during wakefulness to promote sleep. Delivering it locally via flicker avoids the cardiovascular side effects of systemic adenosine administration.
Why 40 Hz specifically and not other frequencies?#
The frequency dependence appears tied to neuronal energy metabolism. Forty-hertz stimulation drives cortical neurons through AMPK-associated metabolic pathways that generate intracellular adenosine, which is then exported via ENT2 [3]. Twenty Hz and 80 Hz do not trigger sufficient adenosine release. The 80 Hz condition showed some analgesic activity in the pain study but significantly less than 40 Hz, and 80 Hz was ineffective for sleep promotion and ischemic neuroprotection [2][3].
Can 40 Hz light flickering replace pain medication?#
Not based on current evidence. All pain data comes from mouse models with sample sizes of 6–16 per group. While the mechanistic story is strong — circuit necessity and sufficiency demonstrated, molecular pathway delineated — no human pain trial has been published. The sleep study did include a small human component (children with insomnia), showing flicker promoted sleep [3], but the pain application remains entirely preclinical. I would not recommend replacing any prescribed pain regimen with flicker therapy at this stage.
Who should avoid 40 Hz light flickering therapy?#
Anyone with photosensitive epilepsy, a history of seizures, or photic-triggered migraines should avoid this without neurological clearance. Pregnant individuals and children should also exercise caution given the absence of safety data in these populations. The flicker frequency range of 15–60 Hz overlaps with the photosensitive seizure threshold for susceptible individuals.
VERDICT#
Score: 7.5/10
The mechanistic work here is genuinely excellent — circuit-level sufficiency and necessity, identified neurotransmitter, specific receptor subtype, convergent evidence across three publications from the same group. This is how preclinical neuromodulation research should be done. The problem is exactly that: it's preclinical. All pain data is from mice. The sample sizes in optogenetic experiments are small (n=6). The "pain memory erasure" claim needs stronger molecular verification. And the same research group has produced all three converging adenosine papers — I want independent replication.
The honest assessment: this is among the most mechanistically convincing arguments for light-based pain therapy I've seen, and it still isn't ready for clinical recommendation. If human trials confirm even half of these effects, this could reshape non-pharmacological pain management. Until then, it's high-quality preclinical science that deserves attention, not prescription.
References
- 1.Author(s) not listed. 40 Hz light flickering alleviates chronic pain via adenosine signaling in the retina-amygdala pathway. Cell Research (2026). ↩
- 2.Author(s) not listed. 40 Hz flicker preconditioning protects nonarteritic anterior ischemic optic neuropathy via adenosine signaling. Communications Biology (2026). ↩
- 3.Author(s) not listed. 40 Hz light flickering promotes sleep through cortical adenosine signaling. Cell Research (2024). ↩
Sova Reld
Sova writes with focused intensity and low tolerance for vague claims. She came to photobiomodulation through personal experimentation and is irritated by both true believers and reflexive skeptics. Her writing has edge: 'The wellness market has done more damage to this field than the skeptics ever could.' She's extremely precise about parameters — wavelength, irradiance, duration — and will tell you when a study used inadequate dosing without apology.
View all articles →