Photobiomodulation for Diabetic Neuropathy Pain: Evidence Review
SNIPPET: Photobiomodulation therapy (PBMT) shows significant promise for reducing neuropathic pain and improving nerve conduction in diabetic peripheral neuropathy. Multiple systematic reviews of randomized trials confirm analgesic effects via mitochondrial activation and cytokine modulation, with low adverse event rates — though protocol standardization remains the field's critical unresolved problem.
Photobiomodulation for Diabetic Neuropathic Pain: What the Latest Systematic Reviews Actually Show
THE PROTOHUMAN PERSPECTIVE#
Diabetic peripheral neuropathy affects an estimated 50% of people with diabetes over their lifetime. That's not a fringe condition. It's a mass-scale degradation of the somatosensory system — numbness, burning, balance loss, and eventually ulceration and amputation. The pharmacological toolkit for this is, frankly, inadequate. Gabapentinoids dull the signal but hammer cognition. Opioids are a dead end. So when multiple systematic reviews land in the same quarter pointing toward a non-pharmacological intervention that modulates mitochondrial efficiency and neural repair pathways with near-zero adverse events, that deserves serious attention. Photobiomodulation isn't new. But the convergence of evidence in 2026 — including a novel laser shoe device and two independent systematic reviews — represents a genuine inflection point for anyone tracking non-drug pain interventions. For the performance-optimization community, the implications extend beyond diabetic populations: if PBMT reliably modulates nerve conduction velocity and plantar pressure dynamics, it has downstream relevance for proprioception, gait efficiency, and injury recovery in otherwise healthy humans.
THE SCIENCE#
What Is Photobiomodulation Therapy?#
Photobiomodulation therapy (PBMT) — formerly called low-level laser therapy (LLLT) — is the application of red (600–700 nm) or near-infrared (NIR, 700–1100 nm) light to biological tissue at non-thermal intensities. The primary chromophore is cytochrome c oxidase (CCO), the terminal enzyme in the mitochondrial electron transport chain. When NIR photons are absorbed by CCO, the result is a measurable increase in mitochondrial membrane potential, ATP synthesis, and downstream signaling cascades including nitric oxide release and reactive oxygen species modulation[3].
This isn't speculative photochemistry. The mechanism has been characterized across multiple cell types and validated in preclinical neuropathy models including chronic constriction injury, spared nerve injury, and streptozotocin-induced diabetic neuropathy[3].
But here's where it gets complicated. Mechanism clarity does not equal protocol clarity. And that's the central tension in this entire field.
The Korada et al. Systematic Review#
The 2026 systematic review by Korada, Arora, Maiya, and colleagues at the Centre for Diabetic Foot Care and Research (Manipal Academy of Higher Education) specifically examined PBMT's effectiveness on three outcomes in diabetic peripheral neuropathy: neuropathic pain, nerve conduction velocity, and plantar pressure distribution[1].
Plantar pressure distribution is the underappreciated variable here. Most PBMT research fixates on pain scores — and those matter. But abnormal plantar pressure is the biomechanical precursor to diabetic foot ulceration. If PBMT can normalize pressure profiles, it's not just managing symptoms; it's intervening upstream of tissue breakdown.
The review, published in Current Diabetes Reviews, synthesizes evidence across multiple RCTs and concludes that PBMT demonstrates positive effects on pain reduction and nerve conduction parameters. The inclusion of plantar pressure as an outcome variable distinguishes this review from earlier work that treated PBMT as purely analgesic.
I'm less convinced by how much we can extract from a systematic review that pools studies with wildly heterogeneous dosing parameters — but more on that below.
The Laser Shoe: A Novel Delivery Mechanism#
Maiya et al. developed and tested a purpose-built laser shoe device designed to deliver PBMT to the entire plantar surface simultaneously[2]. This is a meaningful engineering step. Traditional PBMT application to the foot involves point-source laser probes applied sequentially to specific anatomical landmarks. That approach misses tissue between treatment points and introduces operator variability.
The laser shoe addresses both problems. By housing multiple laser diodes within a wearable shoe form factor, irradiance is distributed across the full plantar region during each session. The study, published in Current Diabetes Reviews (Volume 21, Issue 3, 2025), evaluated the device in type 2 diabetes patients with peripheral neuropathy, measuring neuropathic pain scores and plantar pressure profiles[2].
The concept — turning a clinical laser protocol into a wearable — is exactly the direction this field needs. Whether the specific device delivers adequate irradiance uniformly across varying foot morphologies is a question I'd want answered with thermographic and dosimetric mapping data, which the available abstract doesn't fully address.

Mechanistic Underpinnings: Beyond Simple Analgesia#
The Martins, Rocha, Watkins, and Chacur review in Frontiers in Photonics (December 2025) provides the deepest mechanistic synthesis available[3]. The key pathways:
- Mitochondrial activation via CCO: Photon absorption dissociates inhibitory nitric oxide from CCO, restoring electron transport chain efficiency and boosting ATP output. In neuropathic tissue where mitochondrial dysfunction drives axonal degeneration, this is the primary therapeutic lever.
- Cytokine modulation: PBMT downregulates pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) while upregulating anti-inflammatory mediators. In diabetic neuropathy, where chronic low-grade inflammation accelerates nerve fiber loss, this shift matters.
- BDNF upregulation: Brain-derived neurotrophic factor supports neuronal survival and axonal regeneration. Preclinical NP models consistently show PBMT increases BDNF expression in dorsal root ganglia and spinal cord tissue[3].
- Oxidative stress reduction: PBMT modulates superoxide dismutase and catalase activity, reducing the oxidative burden that damages Schwann cells and myelin sheaths.
The review also flags something most PBMT advocates ignore: sex-based response differences are poorly studied. Most preclinical NP models use male rodents. The handful of studies including female subjects suggest different pain thresholds and inflammatory profiles that may alter PBMT efficacy. This is a real gap.
The Chronic Pain Systematic Review#
Oliveira, Santos, Pereira, and colleagues published a systematic review in Frontiers in Integrative Neuroscience (February 2026) covering 14 RCTs across multiple chronic pain populations — fibromyalgia, peripheral neuropathies, orofacial pain, and musculoskeletal conditions[4].
The headline finding: most trials demonstrated significant pain reduction with PBM, particularly in fibromyalgia and neuropathy. Some studies also showed functional gains and improved quality of life. Adverse event incidence was low across all included trials.
The honest problem, though, and the authors say so directly: heterogeneity of technical parameters compromises standardization. Wavelength, irradiance, energy density, treatment duration, number of sessions — these varied enormously across the 14 studies. This makes meta-analytic pooling borderline meaningless in some cases. You can't average a 4 J/cm² protocol at 808 nm with a 30 J/cm² protocol at 632 nm and call the result "PBM efficacy." Those are fundamentally different interventions.
Wavelength matters. Irradiance matters. Time matters. Tissue type matters. Most of these studies get at least one of those parameters suboptimal.
COMPARISON TABLE#
| Method | Mechanism | Evidence Level | Cost | Accessibility |
|---|---|---|---|---|
| PBMT (clinic-based) | Mitochondrial CCO activation, cytokine modulation, BDNF upregulation | Multiple RCTs, 2 systematic reviews (2025–2026) | $50–150/session | Requires trained clinician, specialized equipment |
| Laser Shoe (wearable PBMT) | Full plantar irradiation via embedded diode array | Single RCT (Maiya et al., 2024) | Device cost TBD (prototype stage) | Home use potential; not yet commercially available |
| Gabapentin/Pregabalin | Voltage-gated calcium channel modulation | Extensive RCT base, Cochrane reviews | $10–50/month (generic) | Widely available; cognitive side effects common |
| Duloxetine (SNRI) | Serotonin-norepinephrine reuptake inhibition | FDA-approved for DPN, multiple RCTs | $15–200/month | Widely prescribed; GI and withdrawal side effects |
| Capsaicin 8% patch | TRPV1 receptor desensitization | RCTs with moderate evidence | $200–400/application | Clinic application required; localized burning |
| Transcutaneous electrical nerve stimulation (TENS) | Gate control theory, endorphin release | Mixed evidence in DPN | $30–100 (device) | Home use; variable protocols |
THE PROTOCOL#
For individuals with diabetic peripheral neuropathy exploring PBMT as an adjunct (not replacement) to existing care, based on the current evidence:
1. Get baseline nerve conduction and pain assessment. Before starting any PBMT protocol, obtain nerve conduction velocity testing and a validated neuropathic pain score (DN4 or NPSI). You need quantifiable baselines or you're flying blind.
2. Identify appropriate wavelength and device. The evidence supports NIR wavelengths in the 808–850 nm range for peripheral nerve targets. Red wavelengths (632 nm) have weaker tissue penetration and are better suited to superficial applications. If your device doesn't specify wavelength, irradiance (mW/cm²), and spot size — don't use it.
3. Target energy density of 4–8 J/cm² per treatment point. This range appears consistently in the positive-outcome studies. Higher is not better — the biphasic dose response (Arndt-Schulz curve) means excessive energy density can inhibit rather than stimulate cellular activity. Calculate treatment time from your device's power output.
4. Treatment frequency: 3 sessions per week for 4–6 weeks initially. Most positive RCTs used protocols in this range. Daily treatment has not been shown to be superior and may increase the risk of exceeding optimal dose windows.
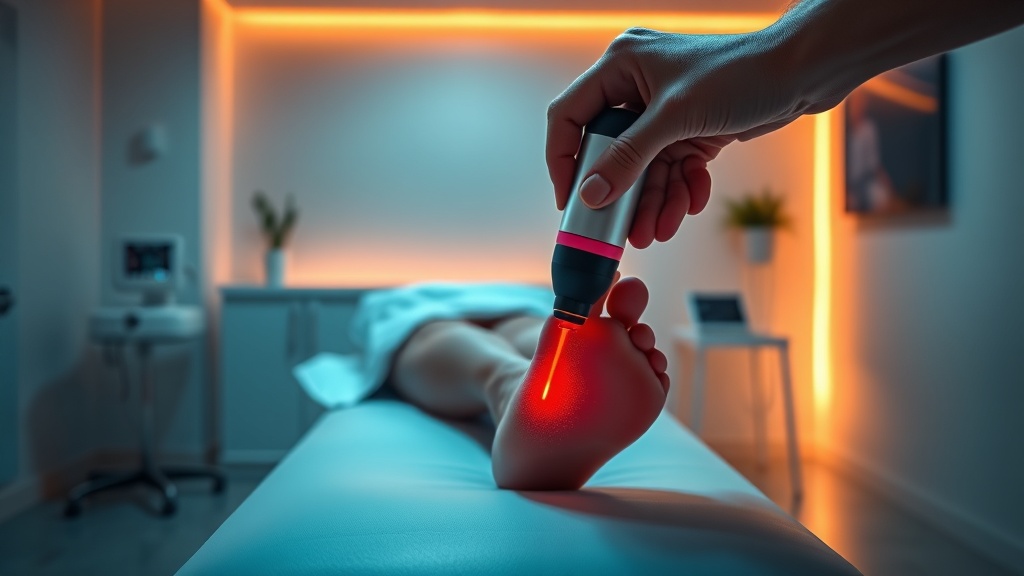
5. Treat the full plantar surface, not just symptomatic areas. The Maiya et al. laser shoe research highlights that comprehensive plantar coverage may be more effective than targeted point application. If using a probe, systematically cover the forefoot, midfoot, and heel in a grid pattern.
6. Track outcomes at 2-week intervals. Re-assess pain scores and, if possible, plantar pressure profiles every 2 weeks during the treatment course. If no measurable improvement by week 4, the protocol parameters may need adjustment — or PBMT may not be the right intervention for that individual.
7. Combine with standard care. PBMT is adjunctive. Do not discontinue prescribed neuropathy medications without medical guidance. The strongest evidence positions PBMT alongside conventional therapy, not as a standalone replacement.
Related Video
What is photobiomodulation therapy and how does it work for neuropathic pain?#
PBMT uses red or near-infrared light at specific wavelengths to stimulate mitochondrial cytochrome c oxidase, increasing ATP production and triggering anti-inflammatory and neuroprotective signaling cascades. In neuropathic pain, this may reduce nerve fiber degeneration and modulate pain signaling at the dorsal root ganglion and spinal cord level. The therapy is non-thermal and non-invasive, with a strong safety profile across published trials[3].
How effective is PBMT compared to standard medications for diabetic neuropathy?#
Based on current systematic reviews, PBMT shows significant pain reduction in most RCTs studying peripheral neuropathy, with very low adverse event rates[4]. However, direct head-to-head comparisons with gabapentin or duloxetine are scarce. The advantage of PBMT lies in its safety profile and absence of systemic side effects, but I'd want to see more comparative effectiveness data before ranking it above first-line pharmacotherapy.
What wavelength and dosage should be used for diabetic peripheral neuropathy?#
The evidence most consistently supports near-infrared wavelengths between 808 and 850 nm at energy densities of 4–8 J/cm² per treatment point[1][3]. The specific irradiance, spot size, and treatment duration will vary by device. Optimal dosing in humans is not yet fully established — this remains an active area of investigation, and the honest answer is that protocol standardization is the single biggest gap in this field.
Why is plantar pressure distribution important in diabetic neuropathy treatment?#
Abnormal plantar pressure is a direct predictor of diabetic foot ulceration, which can lead to infection and amputation. By potentially normalizing pressure profiles, PBMT may address not just the pain symptom but the underlying biomechanical risk factor for tissue breakdown[1][2]. This makes plantar pressure a more clinically meaningful outcome than pain scores alone.
Who should consider photobiomodulation therapy for neuropathic pain?#
Adults with diabetic peripheral neuropathy who have not achieved adequate relief from conventional pharmacotherapy, or who experience intolerable side effects from medications like gabapentin or pregabalin, are reasonable candidates. PBMT should be administered under clinical guidance, ideally with baseline nerve conduction testing. It is not recommended as a standalone treatment — current evidence supports its use as an adjunct to standard care[4].
VERDICT#
Score: 6.5/10
The mechanistic rationale is solid. Cytochrome c oxidase photon absorption is real biochemistry, not wishful thinking. The safety profile is genuinely excellent — across every review examined, adverse events are near-zero. And the convergence of a novel wearable device with multiple systematic reviews in a 6-month window suggests real momentum.
But I can't score this higher until the field solves its protocol problem. The heterogeneity of parameters across studies is not a minor footnote — it's the reason we can't issue definitive clinical guidelines. When one study uses 4 J/cm² at 808 nm and another uses 30 J/cm² at 632 nm and both get lumped into "PBMT for neuropathy," the resulting systematic review tells us less than it should. The laser shoe from Maiya et al. is a smart piece of engineering, but it's a single study. I'd want replication with larger samples and longer follow-up before integrating it into any protocol recommendation with confidence.
The field is moving in the right direction. It's not there yet.
References
- 1.Korada HY, Arora E, Maiya GA, Rao S, Hande M, Shetty S, Gundmi S, Anche P. Effectiveness of Photobiomodulation Therapy on Neuropathic Pain, Nerve Conduction and Plantar Pressure Distribution in Diabetic Peripheral Neuropathy - A Systematic Review. Current Diabetes Reviews (2026). ↩
- 2.Maiya GA, Jadhav RA, Hrishikesh Yadav K, Hande MH, Hebbar H, Mohan K. Effect of Novel Laser Shoe Photobiomodulation on Neuropathic Pain and Plantar Pressure Profile in Type 2 Diabetes Mellitus with Peripheral Neuropathy. Current Diabetes Reviews (2025). ↩
- 3.Martins DO, Rocha IRC, Watkins LR, Chacur M. Photobiomodulation therapy in neuropathic pain: mechanisms, evidence, and future directions. Frontiers in Photonics (2025). ↩
- 4.Oliveira ABC, Santos GN, Ferreira LMA, Mendes JJB, Costa GV, Silva IR, Pereira GS, Silva ML. Photobiomodulation in chronic pain: a systematic review of randomized clinical trials. Frontiers in Integrative Neuroscience (2026). ↩
Sova Reld
Sova writes with focused intensity and low tolerance for vague claims. She came to photobiomodulation through personal experimentation and is irritated by both true believers and reflexive skeptics. Her writing has edge: 'The wellness market has done more damage to this field than the skeptics ever could.' She's extremely precise about parameters — wavelength, irradiance, duration — and will tell you when a study used inadequate dosing without apology.
View all articles →