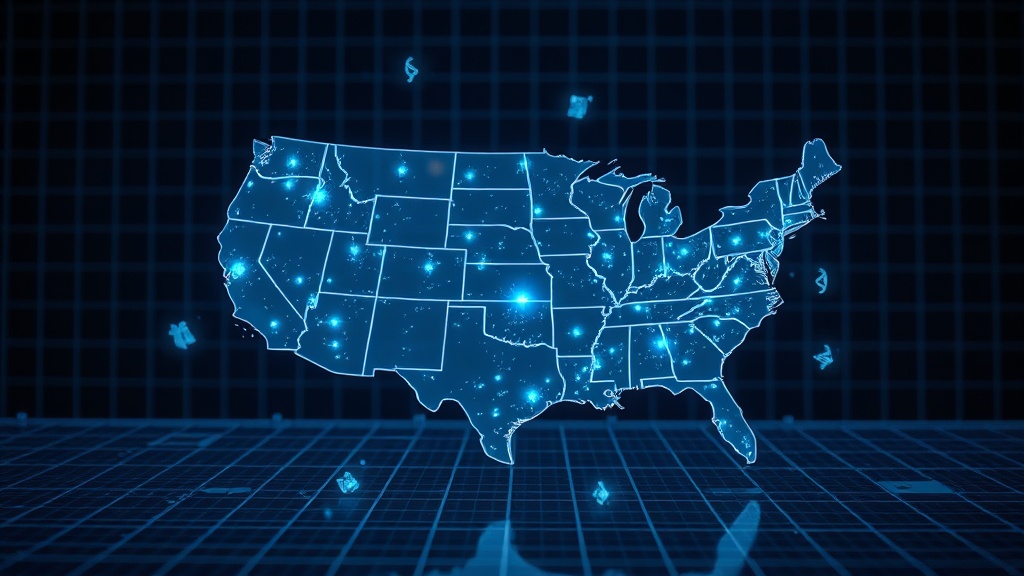
Telomere Length Varies by US Geography and 234 Genetic Loci
SNIPPET: Leukocyte telomere length (LTL) varies significantly by geography, genetics, and ancestry across the United States. A landmark Nature Genetics study of 242,494 participants found longer telomeres clustered on the West Coast and Central Midwest, shorter telomeres in the Southeast, and identified 234 genetic loci — 37 novel — governing telomere dynamics. Your zip code and your genome both write your biological age.
THE PROTOHUMAN PERSPECTIVE#
Telomere length has been the longevity community's favorite biomarker for over a decade, but most of what we knew came from European-ancestry cohorts with average-based measurements. That era just ended. Two studies published in the final week of March 2026 — one in Nature Genetics, one in Nature Communications — have redrawn the map. Literally. We now know that telomere dynamics vary not just by age and lifestyle, but by the chromosome arm you're looking at, the region of the country you live in, your genetic ancestry, and your sex. For anyone optimizing biological age, this changes the calculus. A single average telomere number from a consumer test no longer tells the full story. The data tells me that we've been measuring a shadow on the wall when the actual fire is far more complex — and far more actionable — than we assumed. This matters on a decade-level timescale. This is where longevity science grows up.
THE SCIENCE#
Mapping Telomeres Across a Nation#
Leukocyte telomere length is the measured length of TTAGGG repeat sequences at chromosome ends in white blood cells — a proxy for biological aging at the cellular level. It matters because shorter LTL is consistently associated with cardiovascular disease, pulmonary conditions, and mortality risk, while longer LTL correlates with healthier lifestyle patterns and reduced cardiometabolic burden[1][5].
The numbers from this study are staggering. The All of Us research program analyzed whole-genome sequences from 242,494 participants across diverse ancestries in the United States, then meta-analyzed the results with 679,972 UK Biobank participants — a combined cohort of over 920,000 individuals[1]. That sample size alone makes this one of the largest telomere studies ever conducted.
Here's what the data showed me.
The Geography of Aging#
Significantly longer LTL clustered on the West Coast and Central Midwest, while significantly shorter LTL clustered in the Southeast. This geographic signal persisted after accounting for known confounders. The Southeast — already burdened by higher rates of cardiovascular disease, obesity, and socioeconomic disadvantage — now carries a telomere signature that mirrors those disease patterns[1].
I want to be careful here. The study establishes association, not causation, for the geographic findings. But the alignment between regions with known health disparities and regions with shorter telomeres is difficult to dismiss as coincidence. Lifestyle, socioeconomic status, environmental exposures, and access to healthcare all cluster geographically. The telomere data may be integrating all of those signals into a single biological readout.
234 Loci and the Genetics You Can't Outrun#
The GWAS and meta-analysis identified 234 nonoverlapping loci associated with LTL, of which 37 were entirely novel[1]. Six novel loci were unique to non-European-like populations — a finding that wouldn't exist if this study had followed the tired convention of European-only cohorts. One locus was specific to women.
Rare variant analysis uncovered nine novel genes with functional implications for telomere biology. These aren't just statistical blips; they point toward previously unknown mechanisms of telomere regulation that could eventually become therapeutic targets.
The catch, though. Having 234 loci doesn't mean we understand how they interact. Polygenic scores for telomere length are improving, but the variance explained by genetics alone remains modest. The phenome — lifestyle, biomarkers, socioeconomic status — explained 5.4% of LTL variance in prior UK Biobank analyses[5]. Age and sex account for the majority of that. The genetics add another layer, but the honest answer is that most LTL variance remains unexplained.
Chromosome Arms Are Not Created Equal#
The companion study from Jain et al. in Nature Communications took a different approach — and it's the one that genuinely moved me. Instead of measuring average telomere length, they used long-read whole-genome sequencing on 2,573 All of Us participants to estimate chromosome-specific telomere lengths (csTL)[2][3].
The findings: telomere length varies by chromosome arm, accounting for 9.1% of total csTL variance. Another 8.9% of variance was attributable to the individual, independent of age — supporting the hypothesis that inter-individual differences in telomere length are established at birth and maintained throughout life[2][3].
That second number stopped me. It means that nearly 9% of your chromosome-specific telomere variation was set before you took your first breath. You carry it forward. Age erodes all arms, but longer arms show stronger age-related attrition. And critically, the researchers demonstrated that your shortest individual telomere — not your average — may be the more relevant disease signal[2].
This reframes the entire consumer telomere testing market. An average LTL from a qPCR test cannot detect whether you have one critically short chromosome arm dragging your biology toward dysfunction. The technology to measure this at scale is emerging, but it's not in your direct-to-consumer kit yet.
Measuring Telomeres: The Method Matters#
A separate study comparing DNA methylation-based telomere length (DNAmTL) and qPCR-based measurement found that DNAmTL had superior predictive value for all-cause mortality — AUC of 0.73 versus 0.63 for qPCR[6]. Among 1,601 hypertensive adults followed for a median of 184 months, the highest DNAmTL quartile carried a 36% reduced risk of all-cause mortality compared to the lowest quartile[6].
I'm less convinced by the qPCR comparison here — the sample was relatively small and specific to hypertensive adults — but the direction is consistent: how you measure telomeres matters as much as what you find.
Variance Components in Chromosome-Specific Telomere Length
The Mendelian Randomization Picture#
Prior work using bidirectional Mendelian randomization identified 23 traits that modulate LTL and 24 traits affected by LTL[5]. Smoking cessation and higher educational attainment associate with longer telomeres. Weekly alcohol intake, elevated BMI, high urate levels, and female reproductive events like childbirth associate with shorter LTL. In the other direction, short LTL increases risk for cardiovascular and pulmonary diseases, while longer LTL increases risk for certain autoimmune conditions and cancers[5].
That last point is essential and routinely ignored in biohacking circles. Longer telomeres are not unconditionally better. The Mendelian data shows a U-shaped risk curve — extremely long telomeres are associated with increased cancer risk. The goal isn't maximum length. It's optimal maintenance within a healthy range, with attention to the rate of attrition rather than a single snapshot measurement.
COMPARISON TABLE#
| Method | Mechanism | Evidence Level | Cost | Accessibility |
|---|---|---|---|---|
| qPCR Telomere Length | Measures average relative TL via PCR amplification | Moderate (AUC 0.63 for mortality)[6] | $100–300 | High (consumer tests available) |
| DNAmTL (Methylation-based) | Estimates TL from DNA methylation patterns | Higher (AUC 0.73 for mortality)[6] | $300–600 | Moderate (clinical/research labs) |
| WGS-based LTL (All of Us method) | Estimates LTL from whole-genome sequencing data | High (n = 242,494)[1] | $500–1,000+ | Low (research cohorts) |
| Long-read csTL (Chromosome-specific) | Measures individual chromosome arm TL via lrWGS | Emerging (n = 2,573)[2] | $1,000–2,000+ | Very low (specialized research) |
| Epigenetic Clocks (GrimAge, DunedinPACE) | Multi-biomarker biological age estimation | High (validated for mortality) | $300–500 | Moderate (clinical labs) |
THE PROTOCOL#
Based on current evidence from these studies and prior Mendelian randomization data, here is a practical framework for telomere-conscious longevity optimization.
1. Get a meaningful baseline measurement. If you're serious about tracking biological age, skip basic qPCR telomere tests and opt for DNAmTL through an epigenetic testing service. The data shows it predicts mortality more accurately[6]. Repeat annually.
2. Address the modifiable drivers first. Mendelian randomization identifies BMI, alcohol intake, smoking, and urate levels as causal modulators of LTL[5]. Prioritize body composition optimization, limit alcohol to under 7 drinks per week (or eliminate it), and monitor uric acid levels. These aren't telomere "hacks" — they're the verified upstream levers.
3. Consider your geography and environment. The Southeast clustering of shorter LTL suggests environmental and socioeconomic factors matter[1]. If you live in a region with high pollution, limited green space, or elevated chronic stress, these may be accelerating telomere attrition. Targeted interventions: air filtration at home, regular access to nature, and stress mitigation protocols including HRV-guided breathwork.
4. Optimize NAD+ and mitochondrial pathways — but don't oversell the telomere connection. NAD+ precursors (NMN at 500–1,000 mg/day or NR at 300–600 mg/day) support mitochondrial efficiency and autophagy pathways that may reduce cellular stress contributing to telomere attrition. The direct evidence for NAD+ supplementation lengthening telomeres in humans is limited. I'd frame this as supportive, not primary.

5. Don't chase maximum telomere length. The Mendelian data clearly shows that extremely long telomeres increase cancer risk[5]. The objective is to slow attrition rate, not inflate length. Track rate of change between annual measurements rather than obsessing over a single number.
6. Watch the chromosome-specific space. As long-read sequencing costs decline, chromosome-specific telomere profiling will likely become clinically available within 3–5 years. Your shortest telomere — not your average — may be the critical variable[2]. Early adopters should monitor this technology's progression.
Related Video
What is leukocyte telomere length and why does it matter?#
Leukocyte telomere length is the length of protective TTAGGG repeat sequences at chromosome ends, measured in white blood cells. It serves as a biomarker of biological aging — shorter LTL is associated with increased cardiovascular disease and mortality risk, while the rate of shortening may reflect cumulative lifestyle and environmental exposures over decades[1][5].
How does geography affect telomere length in the United States?#
The All of Us study found that significantly longer LTL clustered on the West Coast and Central Midwest, while significantly shorter LTL clustered in the Southeast[1]. This geographic pattern likely reflects regional differences in socioeconomic conditions, healthcare access, pollution exposure, and lifestyle factors rather than geography alone.
Why are chromosome-specific telomere measurements important?#
Most telomere tests report an average across all chromosomes, but Jain et al. showed that telomere length varies by chromosome arm (accounting for 9.1% of variance) and that an individual's shortest telomere may be more relevant for disease risk than the average[2]. Long-read sequencing now enables this level of resolution, though it's not yet widely accessible.
Who should consider telomere testing?#
Anyone tracking biological age as part of a longevity protocol may benefit, particularly individuals over 40 or those with cardiovascular risk factors. However, DNAmTL-based methods are currently more predictive than standard qPCR tests[6]. I'd recommend pairing telomere data with broader epigenetic clock measurements for a fuller picture.
How many genetic loci influence telomere length?#
The combined GWAS meta-analysis of All of Us and UK Biobank participants (n = 679,972) identified 234 nonoverlapping loci, of which 37 were novel[1]. Six loci were unique to non-European-like populations, underscoring why ancestral diversity in research matters for accurate genomic risk profiling.
VERDICT#
8.5/10. The Nature Genetics study is one of the most important telomere papers published this decade — the sample size, ancestral diversity, and geographic analysis set a new standard. The chromosome-specific companion study is smaller but conceptually transformative. Together, they expose how reductive our previous understanding of telomere biology has been. I dock points because the geographic findings remain associational, and translating 234 loci into actionable clinical tools will take years. But the direction is clear: telomere science is moving from a single number to a multi-dimensional map. That shift matters.
References
- 1.Author(s) not listed. Genomic, phenomic and geographic associations of leukocyte telomere length in the United States. Nature Genetics (2026). ↩
- 2.Jain N, Luo J, Yang Y, Demanelis K, Ahsan H, Aschebrook-Kilfoy B, Chen LS, Pierce BL. Determinants of chromosome-specific telomere lengths among 2573 All of Us participants. Nature Communications (2026). ↩
- 3.Jain N, Luo J, Yang Y, Demanelis K, Ahsan H, Aschebrook-Kilfoy B, Chen LS, Pierce BL. Determinants of chromosome-specific telomere lengths among 2,573 All of Us participants. Nature Communications (2026). ↩
- 5.Demanelis K, Tong L, Pierce BL. Breaking down causes, consequences, and mediating effects of telomere length variation on human health. Genome Biology (2024). ↩
- 6.Author(s) not listed. DNA methylation-based telomere length is more strongly associated with long-term all-cause mortality than quantitative polymerase chain reaction-based telomere length among middle-aged and older hypertensive adults. Clinical Epigenetics (2025). ↩
Orren Falk
Orren writes with the seriousness of someone who thinks about their own mortality every day and has made peace with it. He takes the long view, which means he's less excited than others about marginal gains and more focused on whether something moves the needle on a decade-level timescale. He'll admit when a study impresses him: 'This one actually moved me.' He uses 'the data' as a character in his writing — it speaks, it tells him things, it sometimes disappoints him.
View all articles →
